chemistryworld: “Recent research, led by Brian Cunningham at the University of Illinois in the US, has produced biomedical tubing that uses surface enhanced Raman spectroscopy (SERS) to monitor the contents and concentrations of drugs within a patient’s IV line. The plasmonic nanodome array surface enhances the Raman signals. The tubing could detect 10 pharmaceutical compounds with reproducible signals for a period of up to five days. For four of the drugs, the signal magnitude was dependent upon the drug concentration and combinations of compounds could also be detected, giving a much more detailed picture of a patient’s medication.†– This is great work being done by the University of Illinois. I’ve contemplated something like this in the past.
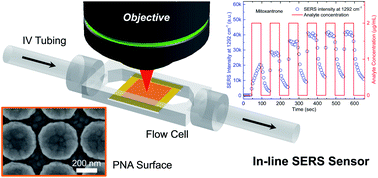
IV drug errors can be devastating. The problem with compounded sterile preparations (CSPs) is that there is currently no way to rapidly verify the contents of the final container. Once an item is compounded in the pharmacy and checked by a pharmacist it’s sent to the nursing unit where it’s administered. It doesn’t happen often, but on rare occasion a CSP will be prepared incorrectly and slip through the verification process; Swiss Cheese Model. This is one of the reasons I think IV room technology is the next important area for pharmacy to attack. Utilizing available technology, i.e. bar code scanning, image-assisted volumetrics, gravimetric analysis, etc will help prevent CSP error from reaching the patient. But on the off chance that they do it only makes sense to have something verify the accuracy of the product before it enters the patient.
Products using this technology are likely years away from being used in routine patient care, but it’s nice to know that it’s in the pipeline.
The article referenced: H-Y Wu and B T Cunningham, Nanoscale, 2014, DOI: 10.1039/c4nr00027g